Monoclonal Antibodies and Multiple Sclerosis
Multiple Sclerosis (MS) is a chronic and unpredictable autoimmune disease that attacks the central nervous system. In those affected, the immune system sends T-cells that attack the brain and spinal cord, leading to inflammation and the demyelination of the nerve cells’ myelin sheath. MS also damages nerve cell bodies in the brain’s grey matter and as the disease progresses cortical atrophy, or shrinkage of the cerebral cortex, may occur. Symptoms of MS vary from loss or doubling of vision, muscle weakness or stiffness, tingling or numbness in the extremities, mental or physical fatigue, depression and cognitive issues (National Institute of Neurological Disorders and Stroke, 2024). There are multiple types of MS that are classified based on length of symptom presentation, reoccurrence and progression of symptoms. Clinically isolated syndrome (CIS) describes symptoms that come from a single attack followed by complete or a near complete recovery and occasionally goes undetected as an MS episode. Relapse-remitting MS (RRMS) symptoms come in recurrent attacks with periods of inactivity called remissions and is the most common diagnosis type. RRMS can become more debilitating with recurrent acute episodes and can be classified as secondary-progressive (SPMS) relapsing or non-relapsing depending on the patient’s presentation. Primary-progressive MS (PPMS) is the least common disease presentation and is characterized by constant progression of worsening symptoms with no remission (National Multiple Sclerosis Society, 2024). Women are more likely to develop MS than men, and the disease is most common in the white ethnicity. While the disease is not hereditary, having a parent or sibling with MS increases the likelihood of development. MS is diagnosed by performing detailed neurological examination and contrast MRI scans to identify the diseases hallmark lesions in the brain or spinal cord. There is no cure for MS, and the primary treatment is corticosteroids to suppress the immune system and inflammatory response. However, corticosteroid treatment has only been proven to quicken the recovery from an MS attack and plays no part in altering the course of the disease (National Institute of Neurological Disorders and Stroke, 2024).
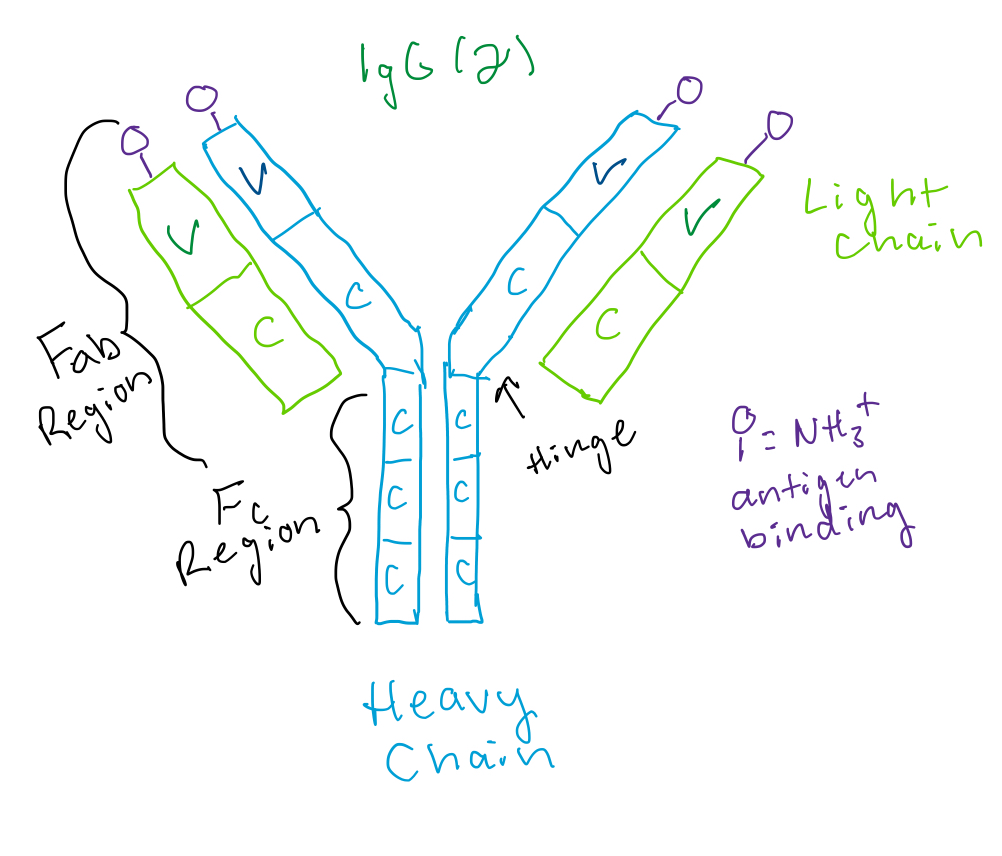
The monoclonal antibody (mAb) drug that I have chosen to discuss is Ofatumumab. Monoclonal antibodies are a newer course of treatment for MS, having received FDA approval in 2004. Ofatumumab has been approved for use in RRMS and works to lessen attacks against the CNS and effectively reduces relapses and lesions caused by inflammation (England, Ferrazzo, Castillo, Klug, & Hoie, 2023). Ofatumumab’s brand name in the United States is Kesimpta and was originally created in 2008 for the treatment of chronic lymphocytic leukemia (CLL) and was approved for use in RRMS patients in 2020. This drug is an anti-CD20 mAb and an IgG1K immunoglobulin type (Drug Bank, 2024). IgG immunoglobulins specifically utilize opsonization to eliminate pathogens, by binding to the target cell’s surface and recruiting phagocytic immune cells and will also activate the classical pathway of the complement system (Stranford, Owen, Punt, & Jones, 2023). Cluster of Differentiation-20 (CD20) is expressed on normal naïve and mature B lymphocytes and is necessary for development and differentiation of B-lymphocytes (InterPro, 2024). The mechanism of action in this drug causes the fragment antigen binding (Fab) region of Ofatumumab’s mAB to bind to CD20 and recruit immunological molecules, such as macrophages, that will mediate immune effector mechanisms such as complement-dependent cytotoxicity (CDC) to promote the destruction of malignant B-cells. While malignancies in B-cells with high expression of CD20 is a result of the leukemia the drug was designed for, it is still somewhat unclear how it provides relief in patients with RRMS, it is presumed to be a consequence of the CD20 binding[1] (Drug Bank, 2024). A common side effect of this drug is an increased susceptibility to upper respiratory infections due to the interference in the patients’ immune system (England, Ferrazzo, Castillo, Klug, & Hoie, 2023).
Works Cited
Drug Bank. (2024, April 23). Ofatumumab. Retrieved from Drug Bank Online: https://go.drugbank.com/drugs/DB06650
England, M., Ferrazzo, A., Castillo, S., Klug, L., & Hoie, E. (2023, March 17). Current Monoclonal Antibody Options for Multiple Sclerosis. Retrieved from U.S. Pharmacist: https://www.uspharmacist.com/article/current-monoclonal-antibody-options-for-multiple-sclerosis#:~:text=In%20MS%2C%20mAbs%20are%20used,inflammation%20that%20results%20in%20lesions.&text=In%202004%2C%20the%20FDA%20approved,mAb%20approved%20for%20this%20ind
InterPro. (2024). B-lymphocyte antigen CD20 . Retrieved from InterPro Classification of Protein Families: https://www.ebi.ac.uk/interpro/protein/reviewed/P11836/
National Institute of Neurological Disorders and Stroke. (2024, September 19). Multiple Sclerosis. Retrieved from NIH: https://www.ninds.nih.gov/health-information/disorders/multiple-sclerosis
National Multiple Sclerosis Society. (2024). Types of MS. Retrieved from National Multiple Sclerosis Society: https://www.nationalmssociety.org/understanding-ms/what-is-ms/types-of-ms
Stranford, S. A., Owen, J. A., Punt, J., & Jones, P. P. (2023). Kuby Immunology. New York: Macmillian Learning.